#"2 e"^(-)"/ orbital" xx "5 orbitals" = "10 e"^(-)#Įach of these ten electrons will have its unique set of four quantum numbers. 
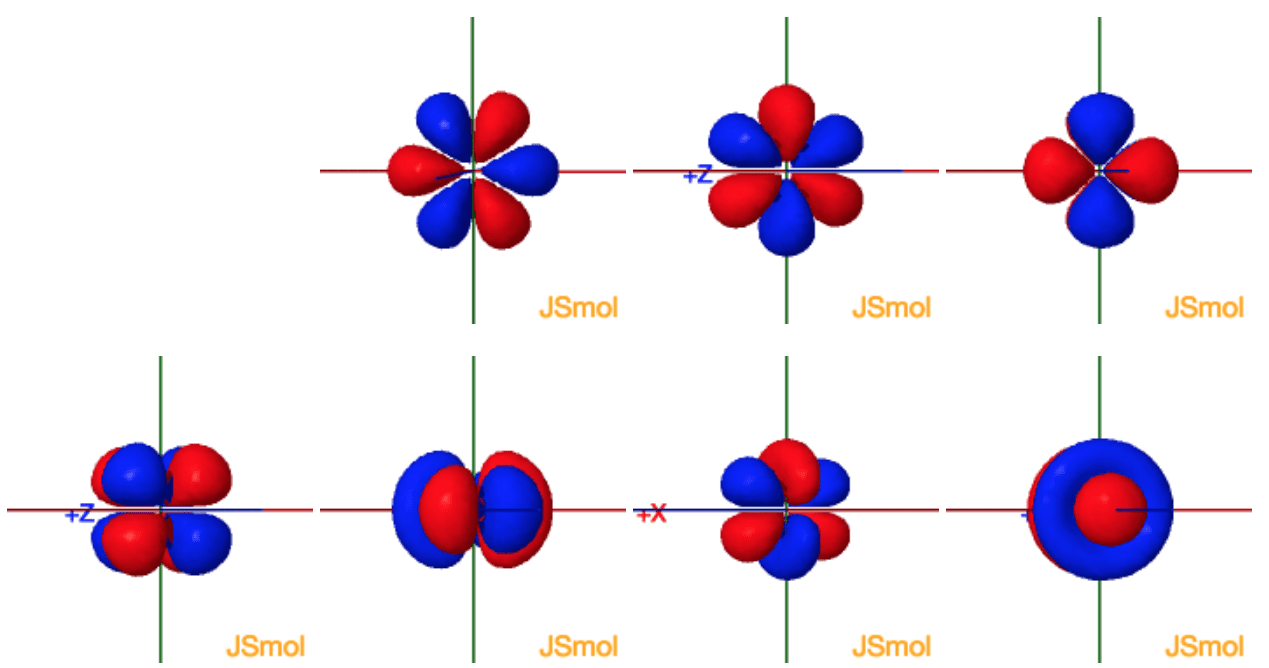
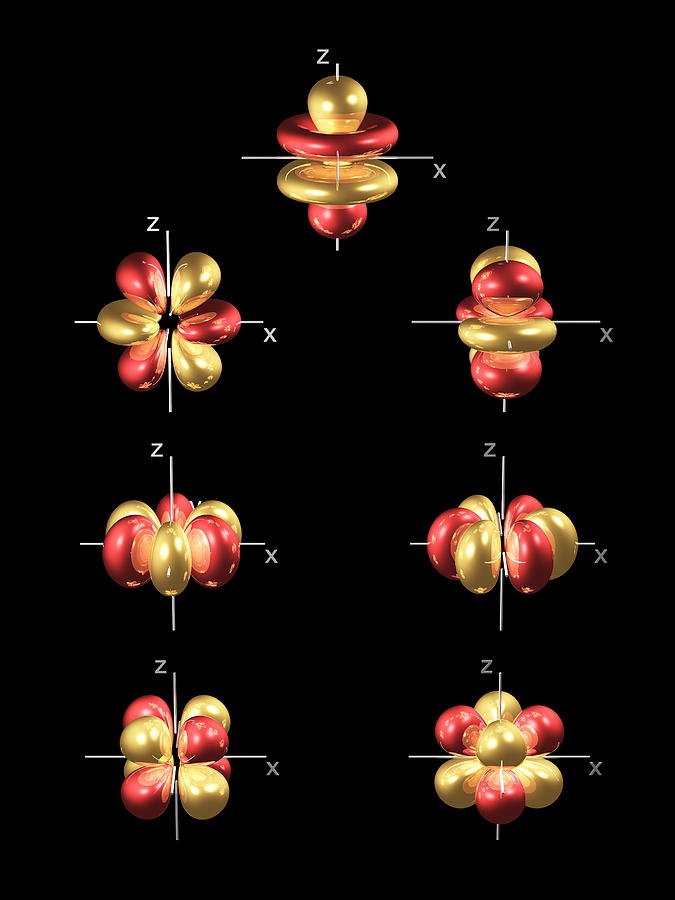
Now, since each orbital can hold a maximum of two electrons, one with spin-up and one with spin-down, it follows that the d-obitals can hold a total of #m_l = #Įach of these five values describes one of the five d-orbitals available in a d-subshell.įinally ,the spin quantum number, #m_s#, can only take two values, #-1/2# for an electron that has spin-down and #+1/2# for an electron that has spin-up. For any d-subshell, the magnetic quantum number can take the values The specific orbital in which the electron is located is given by the magnetic quantum number, #m_l#. Since you're looking for the d-subshell, you will need #l=2#. the effective nuclear charge of the 4d orbital electron in iodine. The subshell in which the electron is located is described by the angular magnetic quantum number, #l#, which for the fourth energy level takes the following values (c) inner electrons shield electrons in a 3p orbital more effec-tively than electrons. #n = color(red)(4) -># the electron is located on the fourth energy level So, the principal quantum number, #n#, describes the energy level on which the electron is located. The 4 fz3 orbital (top row in the image above) has a planar node in the xy plane and two conical nodes with their exes along the z -axis. The 4 f orbitals do not possess any radial nodes. Now, you are given a #color(red)(4)d# orbital and asked to find how many sets of quantum numbers can describe an electron located in such an orbital, or, in other words, how many electrons can occupy a #color(red)(4)d# orbital. In the general set of 4 f orbitals, there are four distinct shapes, each of which possess a number of planar and conical nodes. Gaussians are typically used in molecules with three or more atoms.As you know, we use four quantum numbers to describe the position and spin of an electron in an atom.Įach electron has its unique set of quantum numbers, which means that two electrons can share one, two, or even three quantum numbers, but never all four. Īlthough hydrogen-like orbitals are still used as pedagogical tools, the advent of computers has made STOs preferable for atoms and diatomic molecules since combinations of STOs can replace the nodes in hydrogen-like orbitals. Each such orbital can be occupied by a maximum of two electrons, each with its own projection of spin m s. Alternative to the magnetic quantum number, the orbitals are often labeled by the associated harmonic polynomials (e.g., xy, x 2 − y 2). A generic 4dz2 orbital has n4 and l2.n4 specifies the energy level ,and l. 
Įach orbital in an atom is characterized by a set of values of the three quantum numbers n, ℓ, and m l, which respectively correspond to the electron's energy, angular momentum, and an angular momentum vector component ( magnetic quantum number). >Basic Chemistry >What quantum numbers refer to a 4d orbit. The term atomic orbital may also refer to the physical region or space where the electron can be calculated to be present, as predicted by the particular mathematical form of the orbital. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. In atomic theory and quantum mechanics, an atomic orbital ( / ɒr b ə d l/) is a function describing the location and wave-like behavior of an electron in an atom. radial probability distribution curves of atomic orbitals 1s, 2s, 2p, 3s, 3p, 3d, 4s, 4p, 4d etc., quantum mechanics for IIT JEE, CSIR NET, GATE chemistry. To see the elongated shape of ψ( x, y, z) 2 functions that show probability density more directly, see pictures of d-orbitals below. Each picture is domain coloring of a ψ( x, y, z) function which depend on the coordinates of one electron. The two colors show the phase or sign of the wave function in each region. As a result, 5s orbitals are always filled before 4d orbitals. The shapes of the first five atomic orbitals are: 1s, 2s, 2p x, 2p y, and 2p z.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
